DELPHI GENETICS - DELPHI GENETICS YOUR CERTIFIED CDMO PARTNER FOR GMP PLASMID DNA BIOPRODUCTION
Belgique
Prestataire de services


DELPHI GENETICS
Belgique
With thousands of DNA vectors designed and constructed, Delphi Genetics is a Partner of choice to generate your plasmid of interest. Size does not matter, that's the strategy of cloning that drives the success of a plasmid construction. We perform the construction of your vector from strategy design to final plasmid production. We work with standard plasmid DNA as well as with large constructs, such as BAC and Cosmid. Our plasmid DNA construction service includes: Sequences analysis Design and strategy Gene Synthesis Cloning of PCR fragment DNA fragment assembling Site directed mutagenesis DNA sequencing Plasmid productionA new hope for Covid-19 treatment Delphi Genetics more than triples its production capacity and receives GMP certification. New Plasmid DNA production capacities now immediately available. Delphi to attend Phacilitate Leaders World in Miami, Jan 21-24 Delphi Genetics significantly expands its capacities for cGMP plasmid DNA GMP Certification for Plasmid DNA
Demander un devis
DELPHI GENETICS
Belgique
Delphi Genetics provides customized plasmid DNA production services, from milligram to gram scale, for your R&D and clinical activities. We take care of your plasmid DNA material for viral vectors production and naked DNA-based gene therapy. Our service includes the supply of: R&D grade for non-clinical use, support for lead identification, clone screening, STABY evaluation. High Quality and GMP grade plasmid DNA for viral and non-viral (pre-)clinical activities.
Demander un devis
DELPHI GENETICS
Belgique
GMP grade plasmid DNA production is based on robust, single-use, TSE-BSE-free methods and occurs in our GMP suite, at a maximum scale of 50L. Delphi Genetics offers one-stop-shop CDMO service for plasmid DNA GMP supply, including: GMP compliant Process and QC Development GMP E.coli cell banking (Master and Working Cell Banks) GMP DNA Manufacturing using TSE-BSE free and Single-use process, based on QP approved Master Batch Record QC and Release by Qualified Person (QP) Full Quality Control complying with the European pharmacopea 5.14 will be performed: Content and purity by UV measurement (260/280 nm) Visual inspection Identity by restriction and DNA sequencing (full plasmid sequencing) Safety tests: Endotoxins, Bioburden/Sterility and Mycoplasma Residual RNA content Residual host protein Residual host genomic by Q-QPCR pH Topology Other tests can be performed on request. QP Released GMP Plasmid DNA is provided with TSE-BSE-free certificate and Certificate of Analysis.
Demander un devis
DELPHI GENETICS
Belgique
High-Quality grade plasmid DNA production is based on robust, single-use, TSE-BSE-free methods and occurs in our GMP suite, at a maximum scale of 50L, after Delphi's process performance assessment. Full Quality Control complying with the European pharmacopea 5.14 will be performed: Content and purity by UV measurement (260/280 nm) Visual inspection Identity by restriction and DNA sequencing (full plasmid sequencing) Safety tests: Endotoxins, Bioburden/Sterility and Mycoplasma Residual RNA content Residual host protein Residual host genomic by Q-QPCR pH Topology Other tests can be performed on request. High quality Plasmid DNA is provided with TSE-BSE-free certificate and Certificate of Analysis.
Demander un devis
DELPHI GENETICS
Belgique
R&D plasmid DNA will be produced in shake flask or in fermentor (up to 8L), in our R&D facility, and purified by classical method. R&D plasmids are provided Endotoxin-free.
Demander un devis
DELPHI GENETICS
Belgique
Modern DNA engineering requires efficient and robust molecular tools. The Staby® Operating System, developed by Delphi Genetics is an original set of bacterial strains and vectors integrating unique patented technologies. The use of these products and technologies allows researchers to reach their goal faster and more efficiently. These technologies can be used in a project of any scale (from laboratory scale to industrial processes). The Staby® Operating system is very flexible and its elements can be integrated into your favorite tools, allowing you to add the power of the Delphi-Genetics technologies into your own platform.
Demander un devis
DELPHI GENETICS
Belgique
For manufacturing recombinant proteins or DNA, plasmid instability is a significant concern. Typical biomanufacturing processes in prokaryotes require the use of bacterial plasmids as vectors carrying the gene of interest to be over-expressed. It has been demonstrated that the growth of plasmid bearing cells is significantly reduced relative to plasmid free hosts, simply because protein production represents a significant burden on cellular metabolism. This is of significant concern in the Biopharmaceutical Industry as both the yield and the production reproducibility of recombinant molecules are significantly lowered by that fact. Antibiotic resistance genes are the most common selectable markers used in fermentation processes to avoid plasmid free cells to overgrow the culture. However antibiotics are expensive compounds and they (or their degradation products) can contaminate the biomass or production product.
Demander un devis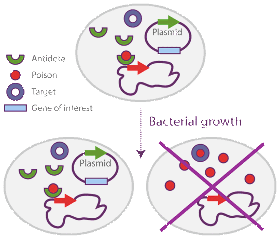
DELPHI GENETICS
Belgique
Several alternative strategies have been developed to minimize the risk that plasmid-free cells overtake the culture. One of the most promising of those strategies relies on the use of postsegretional killing genes (so called poison / antidote genes or selection modules) that induce host killing upon plasmid loss. Delphi Genetics has designed a novel and highly effective stabilization system, called Staby®, based on the use of selection modules naturally found in plasmids, bacterial chromosomes and bacteriophages. A selection module (like the ccd system used by Delphi Genetics) is typically organized as an operon and composed of two genes: the ccdB selection gene codes for a small stable protein which is toxic for E. coli, whereas the ccdA antidote gene codes for a small unstable protein that neutralizes the toxic protein both transcriptionaly and via protein-protein interactions. This unique system allows for the perfect stabilization of the plasmid without the use of antibiotics
Demander un devis
DELPHI GENETICS
Belgique
In order to maximize protein expression, the Staby® system was combined to the proven T7 polymerase technology for the over-expression of recombinant proteins: the gene of interest is under the control of the T7 promoter. This combination is called StabyExpress®. The pStaby1 plasmid contains the gene encoding the antidote protein (ccdA) which counteracts the action of the ccdB protein. The SE1 strain, in which the ccdB selection gene is inserted in the chromosome in order to stabilize the pStaby1 plasmid has been engineered by Delphi Genetics as a dedicated bacterial strain for protein expression and is derived from BL21(DE3). Consequently, IPTG has to be used to induce protein expression, an alternative strategy being the use of an auto-inducible medium (such as Delphi Genetics' Staby®Switch medium). Staby®Express ensures that only plasmid-bearing clones can grow whereas every clone losing the plasmid will inevitably die.
Demander un devis
DELPHI GENETICS
Belgique
Because the stabilization relies on the presence of only two genetic elements, one on the plasmid the other in the expression strain, the stabilization technology is readily adaptable into any existing E.Coli based expression platform. Delphi Genetics can customize any vectors and strains by adding the stabilization cassette into the vector and the selection gene into the chromosome of the chosen expression strain. Furthermore Delphi Genetics has developed a comprehensive licensing policy for those who would wish to integrate the stabilization system into an existing platform.
Demander un devisPrestataire de services
Rue A. de Saint-Exupéry, 5
6041 Gosselies - Belgique
europages vous recommande également
Une sélection d'entreprises proche de l'activité :
Une sélection de produits qui pourraient vous intéresser

ALPHANOSOS
France
Alphanosos vous offre la possibilité d’en découvrir et breveter des nouveaux actifs formés de mélanges synergiques d’ingrédients déjà autorisés dans votre produit ou process. En effet, Alphanosos utilise ses propres algorithmes d’Intelligence Artificielle pour découvrir des actifs avec des effets biologiques forts et ciblés basés sur des mélanges synergiques, donc brevetables, d’ingrédients non-pharmaceutiques, en particulier des extraits botaniques et naturels. Alphanosos a déjà validé son approche pour l’orientation d’activité biologique sur des bactéries, des levures et des cellules humaines. Au-delà de nos solutions pour le contrôle antimicrobien, nous pouvons vous proposer et développer à façon des actifs pour accélérer les croissances en fermenteur et bio-réacteurs, augmenter la production de métabolites, en fait tout ce qu’il vous est possible de mesurer en laboratoire sur l’organisme qui vous intéresse.
Demander un devis
EXTRACTIS
France
Le stade micro-pilote permet d’évaluer techniquement et économiquement les stratégies de fractionnement / purification / modification de la biomasse végétale de votre projet. Les essais sont menés sur des équipements de taille intermédiaire qui permettent de valider les bilans matières de chaque étape du procédé et de vous fournir une information décisionnelle pour réaliser le transfert industriel. L’atelier micro-pilote est équipé d’un panel d’outils technologiques sur toutes les étapes du fractionnement : Extraction/purification modification (10 – 100L) Ultrasons Séparation liquide/solide Technologies membranaires Electrodialyse membranaire Colonne échange d’ions Concentration Séchage par atomisation
Demander un devis
EXTRACTIS
France
Production à Façon Extraits végétaux titrés (extractions aqueuses / solvants) Formes sèche, liquide, macérats… Contrôle analytique et libération des lots Flexibilité / réactivité / productivité Fonctionnement en 2 x 8 permanent Taille des lots : 2 tonnes de matières premières traitées par batch Traçabilité / contrôle Contrôles analytiques (chromatographie, HPIC, HPLC, GC, spectrophotomètre, rhéomètre, viscosimètre…) Libération de lots > conformité (MS, bactériologie, pureté, caractéristiques physico-chimiques…)
Demander un devis
EXTRACTIS
France
Le stade pilote industriel permet de réaliser le transfert industriel d’un procédé, de mener des tests de robustesse sur différents lots de matière première, de valider des coûts de production et de vous accompagner jusqu’à des préséries. L’atelier pilote propose une offre technologique diversifiée de l’extraction de la matière première jusqu’à la mise en forme de l’extrait. Réacteurs 200 L à 15 m3 dont ATEX Réacteur fond filtrant / KiloLab Toutes séparations liquide/solide (plusieurs tonnes/h dont ATEX) Evaporation : concentration (de 400 L à 1 m3/h) Filtrations tangentielles organiques et minérales -> 30 m2 Electrodialyse, chromatographie d’adsorption Sécheurs sous vide 3 tours d’atomisation -> 100 kg/h de capacité évaporatoire
Demander un devisDemande de devis
Créez une seule demande et obtenez plusieurs devis de fournisseurs vérifiés.
- Fournisseurs qualifiés
- Respect de la vie privée
- 100% gratuit
